Top Cancer Drug Companies (2026 Update): Leaders by Revenue, Pipeline, and Growth
The global oncology market is entering a new phase in 2026—defined by immunotherapy dominance, CAR-T expansion, and next-gen targeted therapies. While legacy giants still dominate revenue, earnings reports from Q1 2026 show a clear shift toward innovation-driven growth and aggressive M&A activity.
This updated ranking incorporates:
Latest Q1 2026 earnings data
Oncology revenue trends
Pipeline strength and strategic positioning
Competitive threats (patent cliffs, biosimilars)
1. Merck & Co.

- 2025 First-Quarter reported sales growth of -2% (compared to the same period in 2024) to USD $15.5 Billion
- 2025 Second-Quarter reported sales: USD $15.8 Billion
- R&D Spending 2024: $17.9 billion
- Headquarters: Rahway, New Jersey
Why it still dominates:
Merck remains the global leader largely due to Keytruda, the world’s top-selling cancer drug.
Updated 2026 Outlook
Oncology still contributes the largest share of revenue (~$30B+)
Aggressively pursuing post-Keytruda growth strategies
Latest Developments (2026)
Exploring major acquisitions (e.g., Inhibrx oncology assets) to offset patent expiry risk (Reuters)
Combination therapies (e.g., T-cell stimulators) are improving response rates significantly
Key Insight
👉 Merck is transitioning from single-drug dominance → diversified oncology platform
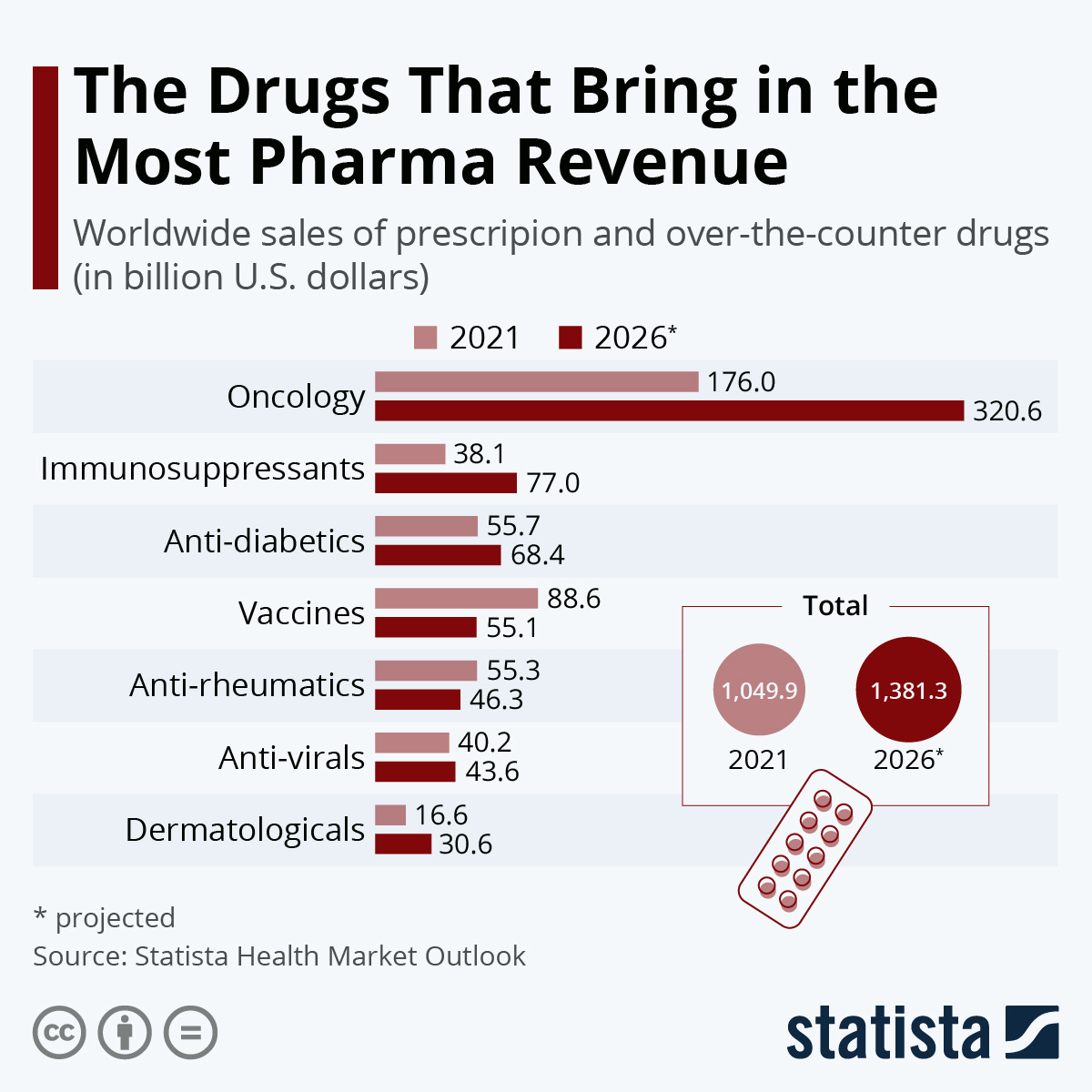
In 2024, Merck's oncology sales were a major driver of its pharmaceutical revenue, with cancer drug sales reaching approximately $32.7 billion. This includes Keytruda (pembrolizumab), Merck's flagship immuno-oncology therapy, which alone generated about $29.5 billion in sales, marking an 18% increase year-over-year. Other oncology products contributing to growth included Welireg, which saw a 133% increase to $509 million, and increased alliance revenues from Reblozyl and Lynparza. Overall, Merck's pharmaceutical sales grew 7% to $57.4 billion in 2024, with oncology being the primary growth area (1, 5, 6, 9).
Keytruda has also impressed in plenty of other cancer types, winning green lights in cancers including head and neck, cervical, lymphoma and more. Its indications across nine collective cancer types will all chip in sales that’ll contribute to its forthcoming leap.
Keytruda

Keytruda is the world's bestselling cancer drug by a large margin. (Merck & Co.)
Company: Merck & Co.
2024 sales: $29.5 billion
Diseases:
- Melanoma, (KEYNOTE-006 and KEYNOTE-054 trials)*
- Non-small cell lung cancer, (KEYNOTE-010, KEYNOTE-407, KEYNOTE-189, KEYNOTE 671 trials)
- head and neck cancer, (KEYNOTE-012 and KEYNOTE-048)
- kidney cancer (renal cell carcinoma), (KEYNOTE-426)
- classical Hodgkin lymphoma, (KEYNOTE-204)
- primary mediastinal large B-cell lymphoma,
- bladder cancer, (EV-302/KEYNOTE-A39)
- microsatellite instability-high or mismatch repair deficient cancers,
- gastric cancer,
- esophageal cancer,
- cervical cancer,
- liver cancer,
- biliary tract cancer,
- Merkel cell carcinoma,
- endometrial cancer,
- tumor mutational burden-high cancer,
- cutaneous (skin) squamous cell carcinoma,
- Triple-negative breast cancer. (ASCENT-04/KEYNOTE-D19 Study)
After AbbVie's Humira lost U.S. market exclusivity and as demand for COVID vaccines plummeted at the end of the pandemic, Merck & Co.’s Keytruda was finally able to claim the title of the world’s bestselling medicine in 2023. But its reign may be short-lived.
In another major feat, a combination of Keytruda and Astellas and Pfizer’s Padcev won full FDA approval as a first-line treatment for advanced bladder cancer—regardless of the patient’s eligibility for chemotherapy. The approval came after the phase 3 EV-302 trial linked the Keytruda-Padcev combo to a 53% reduction in the risk of death compared with chemo.
But the EV-302 readout was viewed as more of a success for Padcev and the antibody-drug conjugate modality, especially given Keytruda’s previous stumbles (Lancet Oncology 2021) in first-line bladder cancer as a monotherapy and as part of a combination with traditional chemotherapy. Nevertheless, the EV-302 regimen speaks to Keytruda’s appeal as the combination partner of choice for newer drugs. Another candidate taking that approach is Moderna and Merck’s individualized cancer vaccine, mRNA-4157 (V940). The shot’s cocktail with Keytruda has drawn much interest because of its promising midphase data in resected melanoma.
However, not all attempts to pair Keytruda up have been successful. Some of the notable failures include Keytruda’s combos with AstraZeneca-partnered Lynparza and Eisai-shared Lenvima.
As Keytruda is slated to fall off the patent cliff in 2028, Merck has been working on a subcutaneous version to potentially soften the blow. However, Merck is behind its rivals BMS and Roche in terms of developing more convenient injections of their PD-1/L1 offerings. A dispute over a microscopic enzyme is threatening Merck & Co. plans to sell a new version of Keytruda, the cancer drug that generates nearly half of the company’s sales. Merck has been tweaking Keytruda to make it easier to use—and to protect billions of dollars in revenue the company could lose after U.S. patent protection runs out in 2028 and rivals can begin selling copycats. The enzyme in the new Keytruda allows it to be injected, rather than given intravenously. It is the subject of a brewing patent dispute between Merck and a biotech called Halozyme Therapeutics. (Wall Street Journal)Some countries (e.g., India, China) may produce biosimilar or off-patent versions of ICIs, but these are not FDA- or EMA-approved and lack global standardization. For example, biosimilars for other biologics (e.g., rituximab) exist in India, but no specific pembrolizumab biosimilar is available in major markets.
2. Roche
- 2025 First-Quarter reported sales growth of 6% to USD $18.7 Billion (CHF 15.4 billion)
- Rank change: None
 |
| New Roche Research Centre (trabantos/Getty Images) |
Legacy leader facing headwinds
Q1 2026 Highlights
Key Products
Ocrevus
Hemlibra
HER2 cancer franchise
Challenges
Biosimilars eating into older cancer drugs
Need for next-gen oncology breakthroughs
Key Insight
👉 Roche is transitioning from legacy biologics → next-gen precision oncology
The lead for the Swiss drugmaker, which has dominated the cancer landscape for years, is about to get a lot less commanding.
One big part of Roche’s forthcoming sales stagnation? Biosimilar rivals, which are readying copies of older blockbusters Rituxan, Herceptin and Avastin. Those copycats are expected to roll out over the next few years, with some already on the market overseas.
To fill the void, Roche is looking in part to immuno-oncology product Tecentriq (checkpoint inhibitor), which currently bears approvals in lung cancer and bladder cancer. There’s just one problem: Its rivals do, too, and analysts say Roche’s data doesn’t seem to stack up to competitor Merck’s in the all-important front-line lung cancer market.
That’s not to say the drug can’t find other markets to control, and recently, trial wins have hinted that Tecentriq may just find some key niches.
Roche is also looking to find new sales avenues for its older drugs through combinations. It’s paired Tecentriq and Avastin in areas such as lung cancer and liver cancer, and it won an FDA approval for a combination of Herceptin and newer breast cancer treatment Perjeta in postsurgery patients, despite data that analysts labeled “weak.”
Then, a few months later, Roche said the closely watched Skyscraper-01 trial of its TIGIT antibody tiragolumab failed to show an overall survival benefit when combined with Tecentriq in first-line PD-L1-high non-small cell lung cancer, despite an earlier positive signal.
Given the importance of the indication, the trial failure pushed the entire TIGIT idea—once billed as the potential next big immune checkpoint target after PD-1/L1—ever closer to its graveyard. Bristol Myers Squibb, Merck & Co. and, most recently, BeiGene, have all ditched TIGIT, although Roche still has a few ongoing phase 3 trials that are fully enrolled.
Also in cancer immunotherapy, Roche last year struck a $1.5 billion deal to purchase its then-partner Poseida Therapeutics, giving the cell therapy field a much-needed injection of confidence. The deal brought to Roche an allogeneic cell therapy platform, which includes a gene editing tool that allows for the delivery of multiple CARs in a single step.
The Poseida buy also strengthened Roche’s flourishing hematology portfolio. Its hemophilia drug Hemlibra grew sales by 12% at constant exchange rates, reaching 4.5 billion Swiss francs last year. Diffuse large B-cell lymphoma (DLBCL) antibody-drug conjugate Polivy crossed the blockbuster threshold with 1.1 billion Swiss francs in 2024. Its two CD19xCD3 bispecifics, Columvi and Lunsumio, are anticipated to receive an FDA decision and a phase 3 readout, respectively, in second-line DLBCL this year.
Overall, Roche has successfully navigated the scary losses of exclusivity of its former top-selling cancer drugs—Avastin, Herceptin and Rituxan. In 2024, group sales went up 3% to about 60.5 billion Swiss francs, including 46.2 billion Swiss francs from the pharma division, which ginned up 4% year-over-year growth.Roche also bought a portfolio of CDK (Cyclin-Dependent Kinase) inhibitors* from China’s Regor Pharmaceuticals last year for $850 million upfront. What’s more, two important phase 3 trials of the company’s oral SERD, giredestrant, could read out this year.
3. Bristol-Myers Squibb

- 2025 First-Quarter reported a total revenue of USD 11.2 B. 6% decrease year over year.
- Rank change: None
- Market Capitalisation: 98.5 B USD
Strength: Deep oncology portfolio led by:
Opdivo
Revlimid (declining due to generics)
CAR-T therapies
Current Situation (2026)
Revenue pressure from Revlimid patent cliff
Growth driven by:
CAR-T therapies (Breyanzi, Abecma)
Next-gen immunotherapies
Key Insight
👉 BMS is shifting from legacy blockbusters → advanced cell therapy ecosystem.
- Bristol Myers Squibb’s Opdivo (nivolumab) is an immunotherapy drug that is used to treat a number of different cancers. Opdivo doesn’t come close to its rival Merck & Co’s immunotherapy Keytruda in terms of sales, but the drug still generated sales of $9 billion in 2024. Thanks to some new indications, Opdivo has seen impressive growth in the last few years, with sales predicted to reach $11.75 billion by 2026. However, BMS cannot afford to rest on its laurels, and they are set to lose the US market exclusively for Opdivo in 2028. Sydney-based company NeuClone Pharmaceuticals, Swedish firm Xbrane Biopharma and China’s Luye Pharma are already working to develop biosimilars. With the loss of exclusivity for not just Opdivo, but also cancer medication Revlimid and Pfizer-partnered blood thinner Eliquis within the next few years, Bristol Myers Squibb is currently working to launch six new drugs, that together could contribute over $15 billion in sales.
4. AstraZeneca

- 2025 First-Quarter Revenue: US$13.6B (up 10% from 1Q 2024)
- Rank change: +2
- Market capitalisation: 225 B USD
Why it stands out:
AstraZeneca has built one of the most balanced oncology pipelines.
Core Growth Drivers
Tagrisso (lung cancer)
Enhertu (explosive growth)
Lynparza (precision medicine)
2026 Position
Oncology = ~40%+ of total revenue
Strong growth in:
Antibody-drug conjugates (ADCs)
Precision oncology
Key Insight
👉 AstraZeneca is the most diversified high-growth oncology company
- Tagrisso, with sales increasing 20% year-over-year to $1.7 billion, primarily for EGFR-mutated non-small cell lung cancer (NSCLC).
- Calquence, used in hematologic malignancies, grew 20% due to new approvals in lymphoma.
- Enhertu, an antibody-drug conjugate, saw a remarkable 48% sales increase, reflecting rapid uptake in HER2-expressing cancers.
- Lynparza, a PARP inhibitor for breast and prostate cancer, posted a 46% revenue surge following expanded indications.
One drug that’ll help lead the charge? Imfinzi, AZ’s entrant in the PD-1/PD-L1 class of immuno-oncology products. The drug won a blockbuster nod in previously treated, stage 3 lung cancer, a sizable market that analysts think it’ll have to itself for years to come.
The biggest splash was from AstraZeneca's drug Imfinzi (durvalumab), which trains a patient's body to attack a protein in their cancer. Imfinzi's already routinely used in some late-stage, recurrent and metastatic cancers (in the treatment of solid lung and liver tumors, for example), but it hasn't been a go-to treatment for earlier-stage cancers.
The results from the company's late-stage phase-3 "Matterhorn" trial (NEJM 2025) presented at the conference, Imfinzi, taken with chemo after surgery, boosted gastric cancer patients' two-year survival rates from 70% (without the immunotherapy) to nearly 76% — a significant jump.
AstraZeneca’s got plenty more going for it in oncology department, though—and it’s been quick to remind investors who are hyperfocused on I-O. For one, it boasts PARP inhibitor Lynparza, which brought in $269 million through the first six months of the year and is riding high on a new breast cancer approval.
Then there’s Tagrisso, a targeted lung cancer drug whose sales grew by 89% year-over-year in H1 to hit $760 million. It’s bolstered by a new indication in first-line EGFR-mutated lung cancer, where it this year became the new standard of care. The EGFR inhibitor king Tagrisso also managed a 13% sales growth to $6.6 billion with two new FDA approvals last year, one in combination with chemotherapy in first-line EGFR-mutated non-small cell lung cancer and the other for stage 3 EGFR lung cancer. But the drug is facing competition from Johnson & Johnson’s cocktail of Rybrevant and Lazcluze.
AZ is also counting on big things from Calquence, expected to challenge Johnson & Johnson and AbbVie juggernaut Imbruvica in blood cancers. While locked in a fierce competition with BeiGene’s rival BTK inhibitor Brukinsa, Calquence was included in the second round of IRA price negotiations.
5. Johnson & Johnson

- 2024 cancer sales: $20.78 billion
- 2025 First-Quarter reported sales growth of 2.4% to $21.9 Billion
- Rank change: +1
- Market Capitalisation: 373 B USD (as of June 2025)
Biggest surprise of 2026 earnings season
Q1 2026 Highlights
Revenue: $24.1B (+9.9% YoY) (Pharmaceutical Technology)
Oncology revenue: ~$6.97B in one quarter alone (Pharmaceutical Technology)
Key Growth Drivers
Darzalex (multiple myeloma)
Carvykti (CAR-T therapy)
Strategic Position
Strong pipeline + diversified pharma portfolio
Raising full-year guidance
Key Insight
👉 J&J is emerging as a top 3 oncology player globally—not just a diversified healthcare company
Johnson & Johnson may not have as wide an oncology portfolio as some of its rivals do, but the products it does have are rock-solid.
J&J is also a force on the multiple myeloma side with TALVEY® (talquetamab-tgvs) and TECVAYLI® (teclistamab-cqyv).
Its Takeda-partnered Velcade long reigned supreme. The standby is still churning out big numbers—it amassed $1.11 billion in 2017 sales outside the U.S., where J&J markets the drug—but it too has a follow-up ready to lead a new generation of myeloma therapies. That’s Darzalex, which by last year had already generated more for the year than its predecessor at $1.24 billion.
Darzalex’s numbers are only set to keep growing, thanks to a landmark FDA green light in May. The agency gave the drug a go-ahead—in combination with Velcade and prednisone—in previously untreated patients who are transplant-ineligible, making Darzalex the first-ever monoclonal antibody to score an OK in newly diagnosed patients.
And then there’s powerhouse Imbruvica, which J&J has shared with AbbVie since the latter’s buyout of Pharmacyclics. That drug pulled in $1.89 billion last year, and a recent combo approval for patients with Waldenström macroglobulinemia should keep sales on the up and up. AstraZeneca, though, is mounting a challenge with Calquence, a drug that’s expected to eventually take on Imbruvica in chronic lymphocytic leukemia.
6. Celgene

- 2024 cancer sales: $18.55 billion
- Rank change: None
Celgene’s oncology portfolio also includes other important therapies, and the company continues to focus on innovative cancer treatments and expanding its market presence worldwide (3).
Celgene Corporation, once a leading biopharmaceutical company focused on cancer and immune-inflammatory diseases and headquartered in Summit, New Jersey, was acquired by Bristol-Myers Squibb (BMS) in late 2019. Following the acquisition, Celgene became a wholly owned subsidiary of BMS, with its stock ceasing to trade and its portfolio integrated into BMS’s operations (1).
Revlimid: The Former Flagship
Revlimid (lenalidomide) long dominated Celgene’s—and now BMS’s—cancer portfolio, particularly in multiple myeloma. However, the landscape has shifted significantly. After reaching peak sales of $12.8 billion in 2021, Revlimid’s exclusivity expired, and generic competition has since entered the market. Teva launched the first U.S. generic in 2025, with several other companies following under volume-limited agreements. These deals gradually increase generic market share until all restrictions lift in 20263.
As a result, Revlimid’s revenue is in decline. BMS projected 2025 sales between $9.5 billion and $10 billion, but expects annual drops of $2 billion to $2.5 billion as generics gain ground3. In the first quarter of 2025, Revlimid generated $936 million, down 44% year-over-year54.
Portfolio Evolution and New Oncology Players
With Revlimid’s decline, BMS has shifted focus to other assets, both legacy Celgene drugs and newer therapies:
Pomalyst: Another multiple myeloma drug, Pomalyst, also faces generic competition in Europe as of August 2024, contributing to a 24% year-over-year decline in Q1 2025 U.S. sales ($658 million)54.
Idhifa: Developed with Agios, Idhifa remains a targeted therapy for relapsed/refractory acute myeloid leukemia (AML) patients with IDH2 mutations. While it was a first-in-class oral inhibitor for this group, its sales are now part of BMS’s broader hematology portfolio.
Vidaza: Vidaza, another AML therapy, continues to contribute but is now categorized among BMS’s mature brands4.
CAR-T Therapies: BMS has advanced CAR-T therapies, notably Breyanzi, which has become the leading CD19-directed CAR-T product in the U.S., driving growth in hematology4.
Reblozyl and Other New Launches: Reblozyl, approved for anemia in beta thalassemia and myelodysplastic syndromes, is now a key growth driver. Alongside it, recent launches such as Breyanzi and deucravacitinib are expected to generate $10–$13 billion in annual sales, offsetting losses from Revlimid’s patent expiry34.
BMS’s oncology and hematology portfolios are transitioning from legacy Celgene blockbusters to a mix of established and newly launched therapies. In Q1 2025, BMS’s growth portfolio outperformed its legacy products, reflecting this strategic shift4. The company is leveraging its broader pipeline and recent acquisitions to sustain growth despite the erosion of Revlimid and Pomalyst revenues (2, 4).
7. Astellas Pharma

- 2024 cancer sales: $12.6 billion (astellas.com)
- Market capitalisation: 17.46 B USD
Strengths
Xtandi (prostate cancer)
Padcev (rapid growth)
Strategy
Focused oncology niches:
Urology
Gastric cancers
Key Insight
👉 Astellas is a mid-tier but fast-growing oncology specialist.
- Xtandi (enzalutamide) for prostate cancer, which showed solid sales growth and remains a cornerstone of Astellas’ oncology portfolio.
- Padcev (enfortumab vedotin) for metastatic urothelial cancer, with sales more than doubling (110% increase) to around ¥117 billion (~$767 million) in the last nine months of 2024.
- Vyloy (zolbetuximab), a therapy for gastric and gastroesophageal junction cancers, which gained momentum due to faster adoption of Claudin 18.2 biomarker testing.
- Xospata (gilteritinib) for acute myeloid leukemia and Izervay (avacincaptad pegol) also contributed to revenue growth.
To summarize, Astellas’ 2024 cancer sales were roughly $12.6 billion, fueled by strong performances from Xtandi, Padcev, Vyloy, and other oncology products, marking it as a significant player in the global oncology market.
8. AbbVie

- 2025 First-Quarter Revenue: US$13.3B (up 8.4% from 1Q 2024).
- Market capitalisation: 335.3 B USD (as of June 2025)
Key Drugs
Imbruvica
Venclexta
2026 Status
Oncology growing but overshadowed by:
Immunology (Skyrizi, Rinvoq)
Key Insight
👉 AbbVie is a secondary oncology player with steady cash flow
9. Novartis

- 2025 First-Quarter Revenue: US$13.2B (up 12% from 1Q 2024)
- Rank change: -4
- Market capitalisation: 251.8 B USD (as of June 2025)
Unique positioning in oncology
Key Strengths
Kisqali (breast cancer blockbuster)
Pluvicto (radioligand therapy)
Strategic Focus
Nuclear medicine / radiopharmaceuticals
High-margin targeted therapies
Key Insight
👉 Novartis is building a dominant niche in radioligand cancer therapy
- Kisqali, a targeted therapy for breast cancer, delivered approximately $3.6 billion in full-year sales (extrapolated from $902 million in Q4 with 52% year-over-year growth), reflecting robust adoption in early and metastatic breast cancer.
- Pluvicto, a radioligand therapy for prostate cancer, generated about $1.4 billion in sales (based on $351 million in Q4 with 28% growth), though it faced some challenges in meeting analyst expectations.
- Scemblix, a tyrosine kinase inhibitor for Philadelphia chromosome-positive chronic myeloid leukemia, received FDA accelerated approval in 2024, contributing to Novartis’ hematologic oncology portfolio.
- Other oncology products such as Leqvio and Fabhalta also contributed to growth, with Fabhalta under priority FDA review for rare kidney disease but relevant to hematologic conditions.
That, of course, was before Merck’s Keytruda and Bristol-Myers Squibb’s Opdivo came along. Both immuno-oncology drugs, now blockbusters, got their start in melanoma before moving into a slew of other indications, and their makers have worked hard to move them earlier into treatment and test them as part of combo regimens.
Since then, Gleevec copies have launched, too, taking a big chunk out of the company’s oncology sales.
That’s not to say Novartis’ cancer sales won’t be growing between now and 2024. They will, thanks in part to the company’s CAR-T therapy Kymriah, which is expected to eventually make a splash, despite early manufacturing woes. Zykadia should be making bigger top-line contributions down the line too, thanks to last year’s front-line ALK+ lung cancer approval.
Novartis is also hoping to get Kisqali going; the HR-positive, HER2-negative breast cancer drug, which competes with Pfizer’s Ibrance and Eli Lilly’s Verzenio, is off to a slower-than-expected start.
By Novartis’ projection, the broad adjuvant nod could lift Kisqali to more than $8 billion in peak sales, versus the $3.2 billion the CDK4/6 inhibitor generated in 2024 mainly from metastatic disease use.
While potential significant growth still lies ahead for Kisqali, the good days of Entresto may be numbered, as Novartis expects the heart failure combo medication will lose U.S. market exclusivity in the coming months. Besides, even if no generics entered this year, Entresto is subject to a price cut under the Inflation Reduction Act beginning in 2026.
Despite the looming Entresto patent cliff, Novartis still expects 2025 sales to grow by mid- to high-single-digit percentages.
From the Medicare Part D reforms, Novartis expects a “modest headwind,” with the biggest impact to come from coverage for the catastrophic phase for Cosentyx and Kisqali in 2025, CEO Vas Narasimhan said during the company’s fourth-quarter earnings call. As to the policies’ potential impact on Novartis’ midterm performance as outlined above, Narasimhan said he’s “very comfortable” with Novartis’ modeling, which takes “appropriately conservative assumptions.”
In two other major expansions for Novartis last year, the FDA granted accelerated approvals to Scemblix in newly diagnosed chronic myeloid leukemia and Fabhalta in the kidney disease immunoglobulin A nephropathy. For both drugs, Novartis has outlined peak sales potential at above $3 billion.
Another potential multibillion-dollar asset, radioligand therapy Pluvicto, also delivered good news for Novartis. With a favorable final overall survival analysis last year from a phase 3 trial, the FDA has in March 2025 approved the PSMA-targeted therapy for metastatic castration-resistant prostate cancer before chemotherapy.
Novartis last year beefed up its radiopharmaceuticals capabilities with the $1 billion upfront acquisition of Mariana Oncology. While Pluvicto uses lutetium as the active substance, Mariana was focused on actinium.10. Eli Lilly — Rapidly Expanding in Oncology
Big move in 2026
Latest Strategic Shift
Acquiring Kelonia Therapeutics (up to $7B) (Investors)
Focus: in vivo CAR-T therapies
Why This Matters
Could eliminate complexity of traditional CAR-T
Positions Lilly against:
J&J
Gilead
BMS
Key Insight
👉 Lilly is transforming from metabolic leader → oncology disruptor.
Key 2026 Industry Trends (From Latest Earnings)
1. Oncology Is Still the #1 Growth Engine
J&J oncology revenue alone nearly $7B in a single quarter (Pharmaceutical Technology)
Most big pharma companies rely on cancer drugs for growth
2. Shift Toward Cell Therapy and CAR-T
J&J, BMS, and Lilly investing heavily
Next wave: in vivo CAR-T (simplified delivery)
3. Patent Cliff Panic (2028–2030)
Merck’s Keytruda expiry is the biggest risk
Industry-wide race for replacement blockbusters
4. M&A Is Accelerating
Final Ranking (2026 Reality Check)
Tier 1 (Dominant Leaders)
Merck
Bristol Myers Squibb
Johnson & Johnson
Tier 2 (High-Growth Challengers)
AstraZeneca
Roche
Novartis
Tier 3 (Strategic Movers)
Eli Lilly
AbbVie
Astellas
Celgene
Bottom Line
The oncology market in 2026 is no longer about who has the biggest drug—it’s about:
Platform innovation (CAR-T, ADCs, radioligands)
Pipeline depth
Strategic acquisitions
👉 The biggest shift from your original article:
Johnson & Johnson and AstraZeneca are rising faster than expected, while Roche and BMS face structural pressure.
Sources and References:
https://www.fiercepharma.com/special-report/top-10-cancer-drugmakers-2024
https://www.fiercepharma.com/special-reports/top-20-pharma-companies-2024-revenue
Disclaimer
The information presented in this article, is intended for general informational purposes only and should not be construed as professional financial, investment, or medical advice. The revenue figures, company rankings, and projections are based on publicly available data, company reports, and industry estimates as of 2025. All currency conversions, where applicable, are based on annual average exchange rates, and revenues outside the health sciences sector have been excluded for consistency (1).
While efforts have been made to ensure the accuracy and timeliness of the information, One Day Advisor and the article’s authors do not guarantee the completeness, reliability, or suitability of the content for any particular purpose. Readers are encouraged to verify details independently and consult qualified professionals before making any business, investment, or healthcare decisions based on the information provided.
The article may reference ongoing developments, regulatory actions, or market events that are subject to change. One Day Advisor is not responsible for any losses or damages arising from the use of this information.
Related:Top 10 most anticipated drug launches of 2025



.png)
.png)

.png)
.png)



Comments